Why Decongestants Can Pose a Cardiovascular Risk
Decongestants are α-adrenergic agonists (sympathomimetics) that constrict blood vessels in the nasal mucosa to relieve symptoms of nasal obstruction.
Decongestants are α-adrenergic agonists (sympathomimetics) that constrict blood vessels in the nasal mucosa to relieve symptoms of nasal obstruction.1
Highly vascularized mucosa covers all surfaces within the human nose.2Exposure to allergens, infections, or irritants, causes dense subepithelial blood vessel network distention, leading to congestion, mucosal swelling, nasal airflow obstruction, reduced nasal cavity volume, and vasodilation.2Decongestants are available in oral and topical intranasal formulations, both of which carry serious cardiovascular risks that warrant clinician attention to patient selection and therapy duration.3
Practitioners must recognize the risks of prescribing and recommending all medications. Effective patient education is important to prevent adverse events (AEs). TheTablesuggests drug-specific counseling points to raise with patients when recommending a decongestant.4
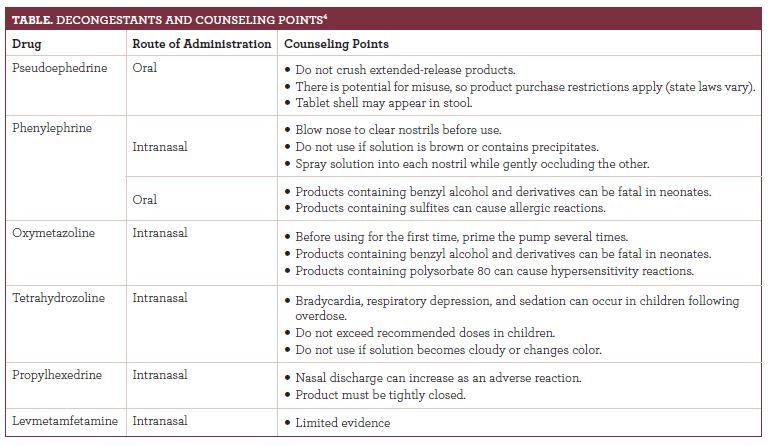
When counseling patients looking for congestion relief, clinicians should determine patient specific risks such as age, comorbidities, concurrent medications, and intended duration.5
Not for Toddlers
The FDA warns that children under 2 years should not receive decongestants because of potential life-threatening AEs, including convulsions and rapid heart rates. In 2008, manufacturers voluntarily revised cough and cold products labels to state “do not use” in children under 4 years.6
Children and older adults are more likely to experience AEs from oral decongestant use.7The most common AEs, insomnia and irritability, can occur in up to 25% of patients.8AEs are more common with oral decongestants because minimal systemic absorption occurs with topical decongestants. Propellant- and vehicle-associated effects include burning, local dryness, sneezing, stinging, and trauma from the tip of the administration device.7Importantly, topical decongestant use can cause rebound congestion or rhinitis medicamentosa. The exact cause is unknown, but long duration of therapy, preservative agents such as benzalkonium chloride, and short-acting products are suspected as contributing factors.7
Adrenergic Stimulation Sensitivity
Most decongestant labels must state, “Do not take this product if you have heart disease, high blood pressure [BP], thyroid disease, diabetes, or difficulty in urination due to enlargement of the prostate gland unless directed by a doctor.”9
Systemic decongestants cause generalized peripheral vasoconstriction,10and AEs include arrhythmias, hypertension, palpitations, and tachycardia.7Hypertension is particularly concerning because it affects nearly half of American adults, with many unaware of their condition.11The American Heart Association recommends that patients with high BP consult a health care professional before taking any OTC medications or supplements.11No clear evidence exists that any 1 decongestant is safest in this population.7
Results from studies show that pseudoephedrine causes an average increase of 1.2 mm Hg in systolic BP in patients with controlled hypertension. However, it is unclear whether this rise in systolic BP is linked to clinical outcomes.12Higher doses and immediate-release preparations were associated with greater BP increases.13
Products intended for patients with high BP (Coricidin HBP) generally do not contain a decongestant. These products usually contain a combination of ingredients that may or may not be appropriate, depending on the patient’s symptoms.7
Interactions Increase Risks
Decongestants can interact with monoamine oxidase inhibitors, tricyclic antidepressants, antihypertensives, indomethacin, β-blockers, methyldopa, and other central nervous system (CNS) stimulants.10
Decongestants effectively reduce respiratory tract obstruction but have no effect on other symptoms, including pruritus, rhinorrhea, and sneezing. Thus, practitioners often recommend decongestants combined with other agents, such as antihistamines and mucolytic agents.3When recommending a decongestant, clinicians must be aware of medication interactions, possible drug-induced causes of nasal congestion, and potential duplicate therapies and overdose risk.10
Rebound congestion from overuse of topical nasal decongestants is the most commonly recognized type of drug-induced congestion. Other drugs that can cause nasal congestion are aspirin, β-blockers, and nonsteroidal anti-inflammatories.10
Recommended Doses, Optimal Durations
It is important to adhere to FDA-approved doses because acute overdoses can be life-threatening. Systemic overdoses cause excessive cardiovascular collapse, CNS stimulation, coma, paradoxical CNS depression, and shock. Symptoms of CNS stimulation include anxiety, fear, hallucinations, insomnia, restlessness, and tremors. If any of these symptoms occurs, patients must discontinue therapy.7
Advise patients to return if symptoms do not improve within 7 days of oral decongestant therapy or are accompanied by a fever.9Always recommend intranasal decongestants for short durations to prevent rebound congestion.3
Remind patients of the importance of self-monitoring BP, especially while taking decongestants. Use open-ended questions to determine how frequently patients monitor their BP, assess their technique, and promote medication adherence.11
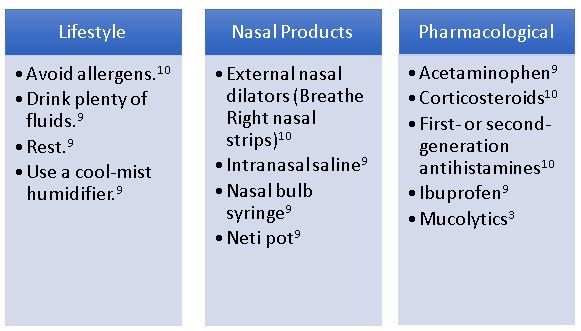
Provide Alternatives to Decongestants9
Clinicians can recommend the lifestyle modifications and nasal products listed in theFigurefor most patients.10Consider the primary symptoms and the underlying etiology of the patient’s condition when selecting pharmacotherapy alternatives or additions to decongestants.5
FIGURE.CONGESTION THERAPY10
Sara E. Miller is a PharmD candidate at the University of Connecticut School of Pharmacy in Storrs.
References
- Gentile DA, Pleskovic N, Skoner DP, et al. Allergic rhinitis. In: Leung D, ed.Pediatric Allergy: Principles and Practice.2nd ed. Philadelphia, PA: Saunders; 2016;210-218.
- Corboz MR, Rivelli MA, Mingo GG, et al. Mechanism of decongestant activity of alpha 2-adrenoreceptor agonist.Pulm Pharmacol Ther. 2008;21(3):449-454. doi: 10.1016/j.pupt.2007.06.007.
- Chadwick SJ. Principles of allergy management. In: Krouse JH, Derebery MJ, Chadwick SJ, eds.Managing the Allergic Patient.1st ed. Philadelphia, PA: Saunders; 2008:19-72.
- Micromedex Solutions. micromedexsolutions.com/home/dispatch/ssl/true. Accessed November 5, 2018.
- Meltzer EO, Caballero F, Fromer LM, Krouse JH, Scadding G. Treatment of congestion in upper respiratory diseases.Int J Gen Med.2010;3:69-91.
- Use caution when giving cough and cold products to kids. FDA website. fda.gov/drugs/resourcesforyou/specialfeatures/ucm263948.htm. Updated February 8, 2018. Accessed November 5, 2018.
- Krinsky DL, Ferreri SP, Hemstreet B, et al.Handbook of Nonprescription Drugs. Washington, DC: American Pharmacist Association. 2018;192-196.
- Corren J. Allergic rhinitis and conjunctivitis. In: O’Hehir RE, Holgate ST, Sheikh A, eds.Middleton’s Allergy Essentials.Milton, ON: Elsevier Canada; 2017:205-224.
- Code of federal regulations title 21. FDA website. accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=341.80. Published September 4, 2018. Accessed November 5, 2018.
- Corey JP, Houser SM, Ng BA, et al. Nasal congestion: a review of its etiology, evaluation, and treatment.Ear Nose Throat J.2000;79(9):690-693.
- The facts about high blood pressure. American Heart Association website. heart.org/en/health-topics/high-blood-pressure/the-facts-about-high-blood-pressure. Accessed November 5, 2018.
- Hollander-Rodriguez JC, Monjoy HL, Smedra B, Prouty JP. Clinical inquiry: do oral decongestants have a clinically significant effect on BP in patients with hypertension?J Fam Pract. 2017;66(6):E1-E2.
- Salerno SM, Jackson JL, Berbano EP. Effect of oral pseudoephedrine on blood pressure and heart rate: a meta-analysis.Arch Intern Med. 2005;165(15):1686-1694. doi: 10.1001/archinte.165.15.1686.

